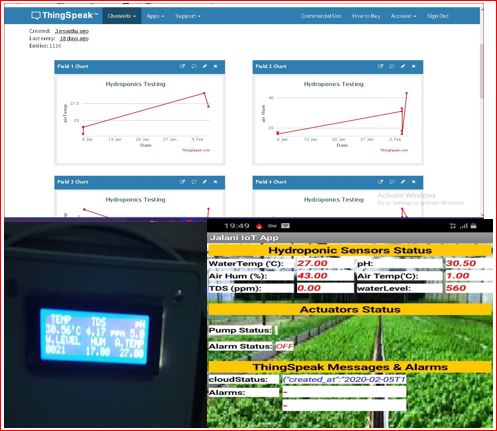
In this situation, “chicken” or at least a specific chicken protein, is the appropriate response. The fact that this specific chicken protein really comes in first and last adds another surprise. Its ingenious approach offers fresh perspectives on how to manage crystal growth, which is essential for the development of eggshells. The cracking of Eggshells is automatically carried out by a well-designed machine to break the eggs with ease. Ovocledidin-17 (OC-17), a protein found in chicken eggshells, was long recognized as having some significance in the development of eggshells. Only the mineral area of the egg—the hard part of the shell—contains the protein, and laboratory experiments revealed that it appeared to affect how amorphous calcium carbonate (CaCo3) crystallizes into calcite. This control’s exact mechanism is yet unknown. Uncertainty persisted on how this procedure would be applied to actually make an eggshell.

The egg problem has finally been resolved by researchers from the University of Warwick, Mark Rodger and David Quigley, together with collaborators from the University of Sheffield. They did this by using the UK national supercomputer in Edinburgh using a powerful computing method called meta dynamics. Using these tools, The Warwick and Sheffield researchers were able to create simulations that showed exactly how the protein bound to amorphous calcium carbonate surface using two clusters of “arginine residues,” located on two loops of the protein and creating a literal chemical “clamp” to nano sized particles of calcium carbonate. This type of clamping, known as the OC-17, promotes the transformation of calcium carbonate nanoparticles into “calcite crystallites,” which are the minuscule crystal nuclei that can continue to grow on their own. However, they also observed that this chemical clamp occasionally failed to function. Only the OC-17 appeared to separate off the nanoparticle or “be desorbed.”

The larger nanoparticles we looked at had the same binding sites for this chemical clamp as the smaller ones, but the binding was significantly weaker, according to Professor Mark Rodger of the University of Warwick’s Department of Chemistry and Centre for Scientific Computing. The protein never fell off or desorbed from the smaller nanoparticle in the simulations we ran, but it did so from the larger one every time. Desorption, though, took place in each instance at or right after calcite nucleation. Thus, the scientists have discovered a remarkably elegant method for the highly effective recycling of the OC-17 protein. In essence, it serves as a catalyst, grabbing onto calcium carbonate atoms to jump-start crystal growth before letting go once the crystal nucleus is big enough to grow on its own. As a result, the OC-17 is free to encourage further crystallization, which enables the rapid, overnight formation of an eggshell. According to the researchers, anyone looking to promote and manage artificial forms of crystallization will greatly profit from this new understanding of the sophisticated and very effective strategies used by nature to do so.